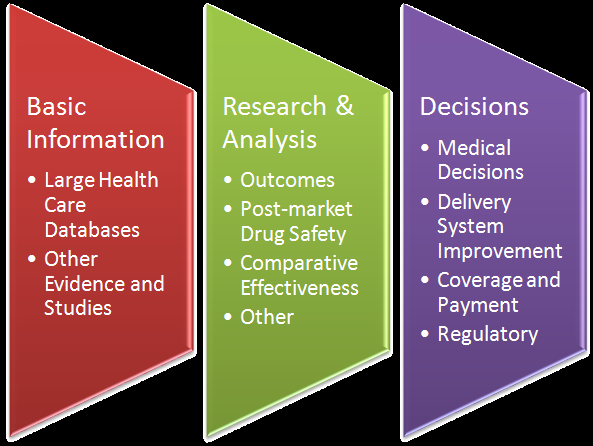
Comparatie Effectivenesss and medical product safety efforts are hot topics. They are related because using large electronic health care data bases will form research or monitoring platforms for each efforts. Nandan Kenkeremath was a lead negotiator on the post-market surviellance and safety provision of the recent Food and Drug Administration Amendment Acts of 2007. He has consulted on the implementation issues associated with this Act and on FDA's sentinel Initiative. Nandan is a consultant for the eHealth Initiative and leads a workgroup on comparative effectiveness research and policy and staffs a collaborative research effort on drug safety. He has been involved in related legislative efforts and prepared the following toolkit for the U.S. Chamber of Commerce.
Click here for Health Care Comparative Effectiveness Toolkit
| 
